pH testing - why & how we do it
NOTE: pH testing is only for formulas which contain WATER. You do not need to pH test formulas which only contain oils, such as oil serums or balms. But as soon as water is introduced get your strips out!
What is pH and why do you have to test it?
pH testing can seem very complicated and overwhelming so here’s a step-by-step guide to get to grips with the basics.
For starters, what is it?
The initials pH stand for "potential hydrogen". It measures the concentration of the hydrogen ions in a solution, which indicates the acidity or alkalinity of any product that contains water. The higher the concentration of hydrogen ions, the lower the pH, the lower the concentration of the hydrogen ions, the higher the pH.
The pH range goes from 0 to 14, with 7 (that of pure water) being neutral. Below 7 indicates acidity and above 7 indicates alkalinity.
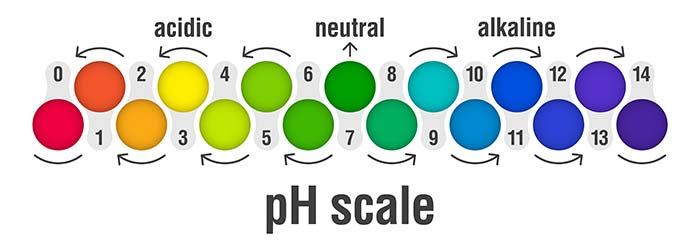
However, our skin is not neutral. Our skin is covered with a protective hydrolipidic layer which makes the optimal pH value of our skin, for our face and most of our body, between 4.7 and 5.75, meaning that it is slightly acidic.
The reason for this slight acidity is to keep moisture in and bacteria out. If skin is too acidic it can increase chances of skin inflammation such as acne or eczema and if it’s too alkaline, skin can appear red and flaky.
So it’s important that the product we make for our skin falls within the optimal skin pH range, which is usually around 5 - 5.5.
The other reason that it is important to monitor the pH of the products you are formulating is because of the ingredients themselves. Some ingredients are not suited to particular pH ranges and may quickly become inactive if the finished product doesn’t fall within their preferred pH range (I’m looking at you vitamin C!).
This includes preservatives - it is very important that they’re doing their job! So to make sure the wonderful actives and functional ingredients in your formula are working to their full potential and ensuring the long term stability of your product, we need to get our pH right.
What you need to test your pH
- pH testing strips. (You can get pH meters, which are more sophisticated and you may wish to invest in one one day, but when you’re just starting out pH testing strips do the job just fine.)
- Distilled water. Always use distilled or deionised water for pH control.
 |
||||
| pH testing strips |
Watch our Formulation 101 - Base Cream video which shows you pH testing in action.
How to test your pH
- Dip the pH strip into your product and check the results against the chart on the packaging.
- You want your product’s pH to sit around 5 - 5.5 (depending on your formula). If that’s where it is then great, you’re good to go.
- If your formula is not within this range then you need to adjust it.
Adjusting the pH with a pH modifier
pH modifiers are ingredients that are added to adjust the pH of your formula. They are usually used in a very small percentage and added in the final steps of the formulation process. You can easily make these up yourself.
pH modifiers to adjust pH UP:
- Sodium hydroxide 10% in 90% distilled water. Example: 1g sodium hydroxide into 9g water. ALWAYS add sodium hydroxide to the water not the other way around or it will create strong fumes. Sodium hydroxide is highly corrosive. Always practice caution when working with it. Cover eyes, hands and clothing.
- Baking soda 10% in 90% distilled water. Example: 1g baking soda into 9g water.
pH modifiers to adjust pH DOWN:
- Citric acid 10% in 90% distilled water. Example: 1g citric acid into 9g water.
- Lactic acid solution. Our lactic acid is 80% strength; you can then dilute it further with distilled water to make 40%, 25% or 10% solutions.
The blended pH modifier solutions can be kept for up to a week in cool, dark place or if you don’t formulate very often, you can make a new batch when you do formulate.
How to use pH modifiers:
- You only need to use a few drops of the pH modifiers to decrease or increase the pH of your product. Add a few drops and make sure it is well combined.
- Once adjusted, check the pH again, repeat if necessary.
Always remember to take notes while you’re formulating. That way when you come to make your favourite formula again you will know what to expect.
Here's a video from The Institute of Personal Care Science on pH testing which is very helpful.
Follow us on Instagram or Facebook for more recipes, news and specials!

Comments